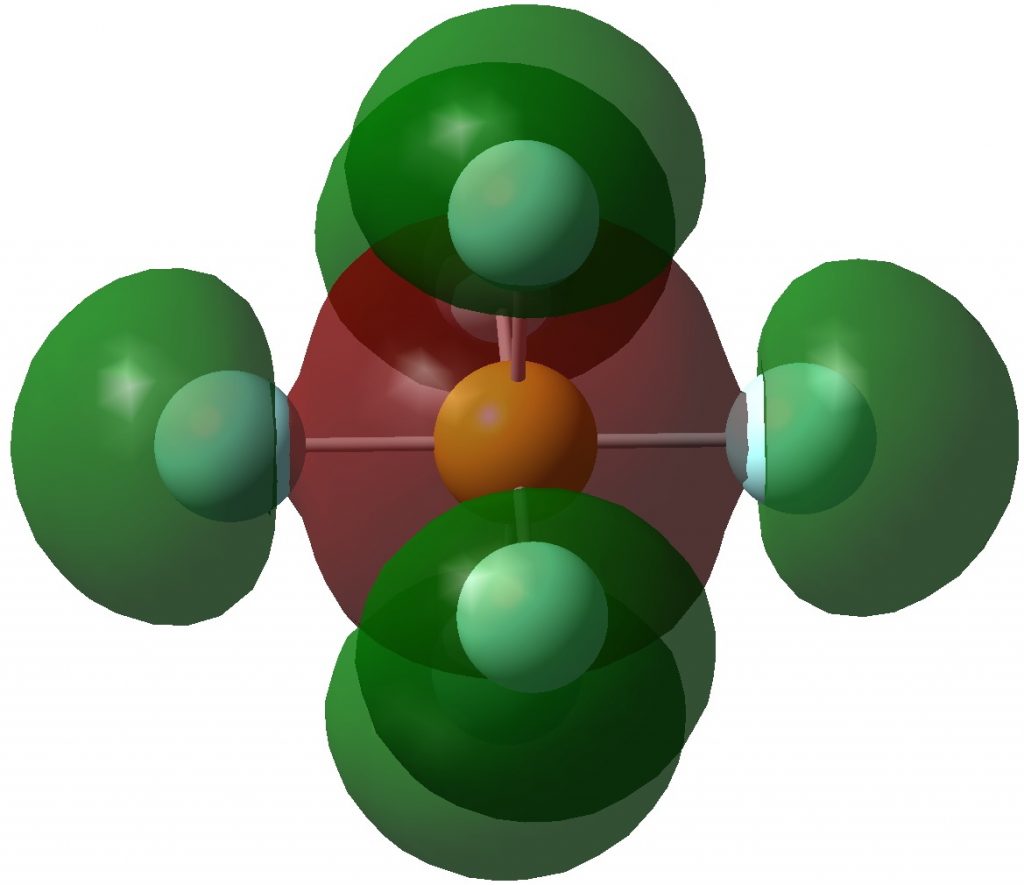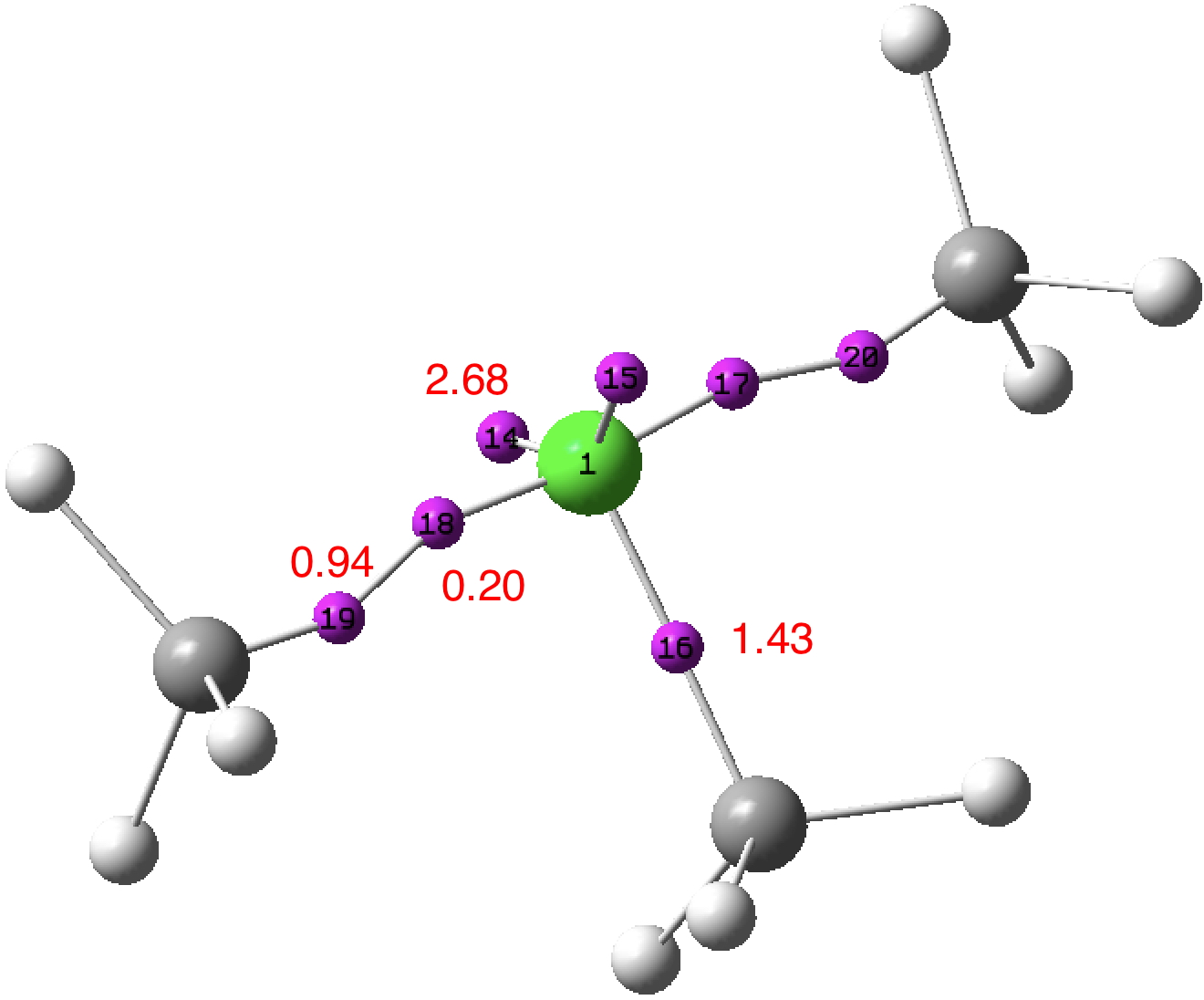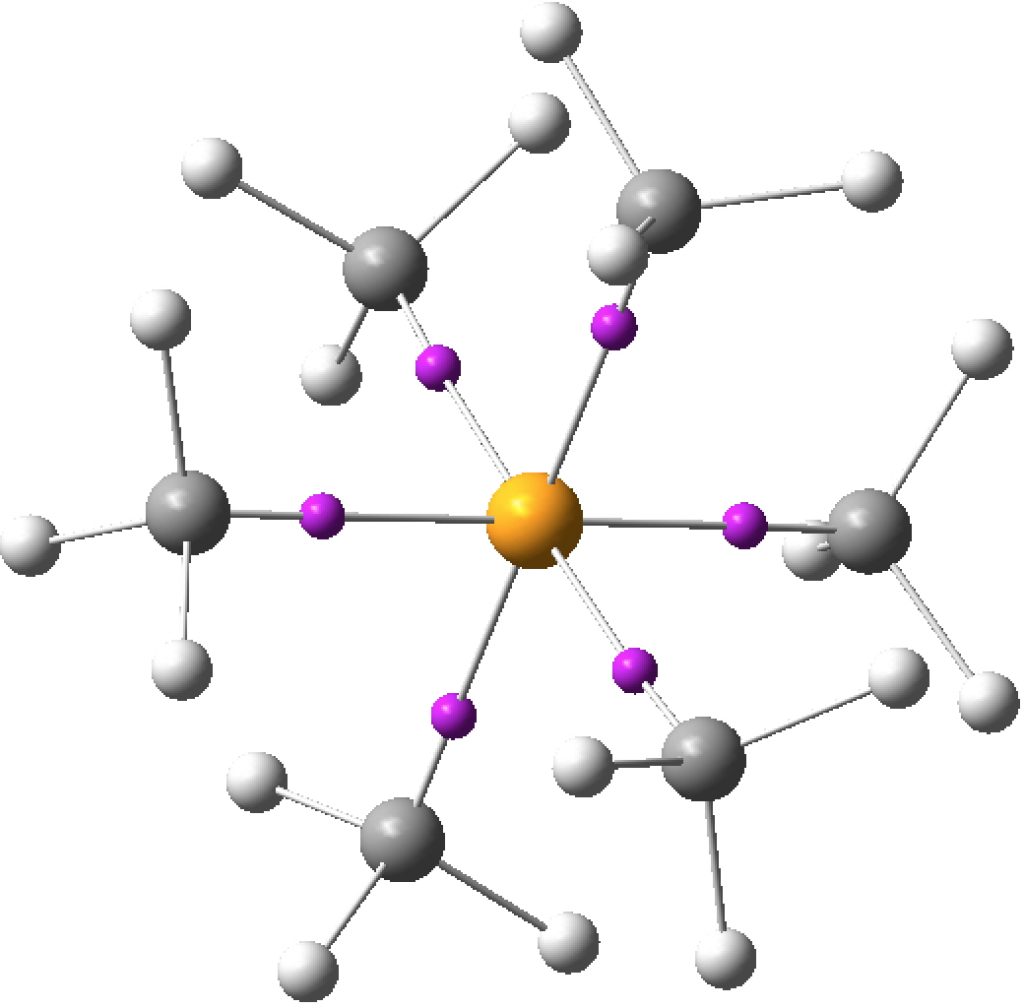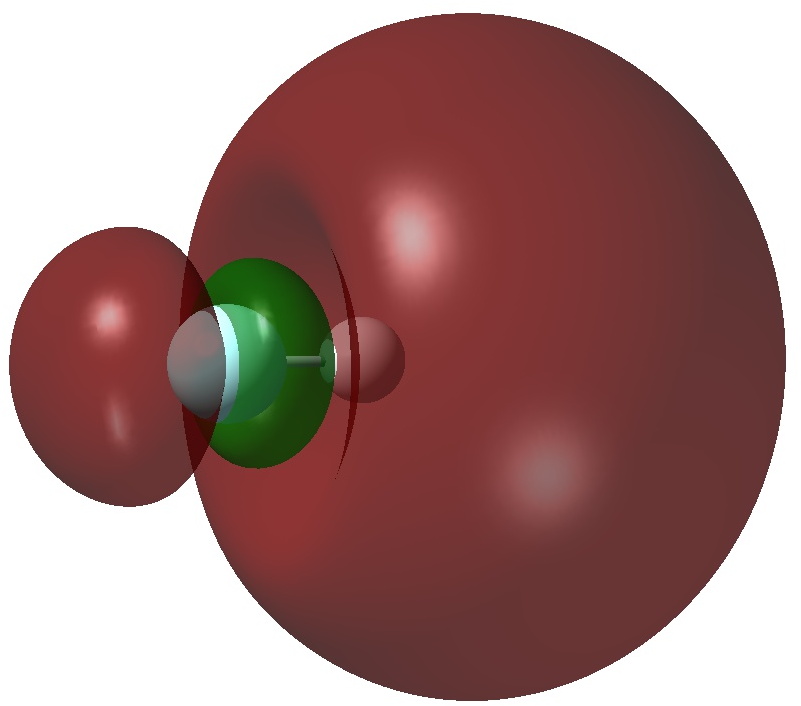
An article with the title shown above in part recently appeared.[cite]10.1038/s41598-017-02687-z[/cite] Given the apparent similarity of HF 1- to CH 3 F 1- and CH 3 F 2- , the latter of which I introduced on this blog previously, I thought it of interest to apply my analysis to HF 1- . The authors[cite]10.1038/s41598-017-02687-z[/cite] conclude that “ the F atom of HF −