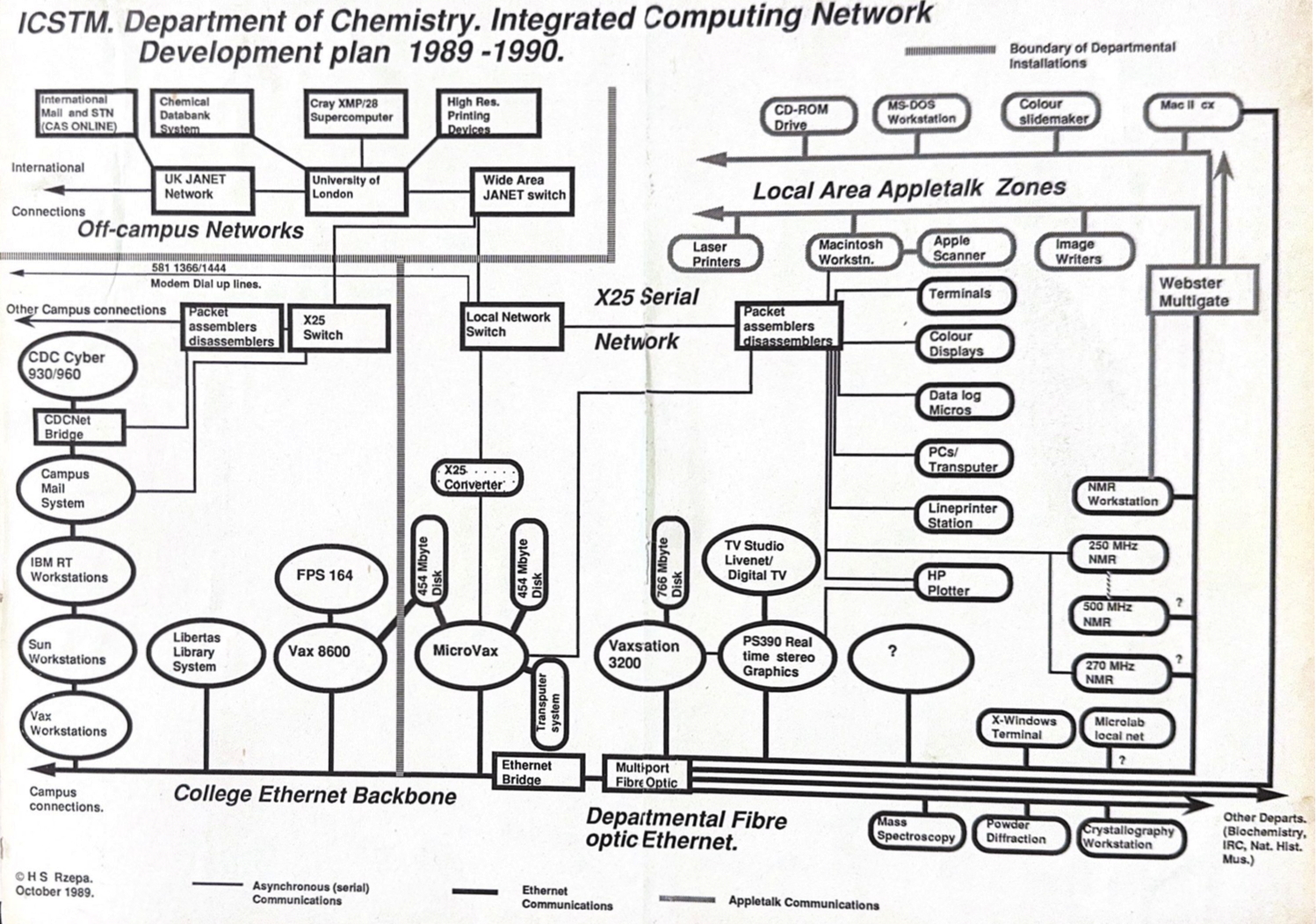
In 2023, we very much take for granted that everyone and pretty much everything is online. But it was not always so and when I came across an old plan indicating how the chemistry department at Imperial College was connected in 1989, I was struck by how much has happened in the 34 years since.