This potential example of a molecule on the edge of chaos was suggested to me by a student (thanks Stephen!), originating from an inorganic tutorial. It represents a class of Mo-complex ligated by two dithiocarbamate ligands and two aryl nitrene ligands (Ar-N:).
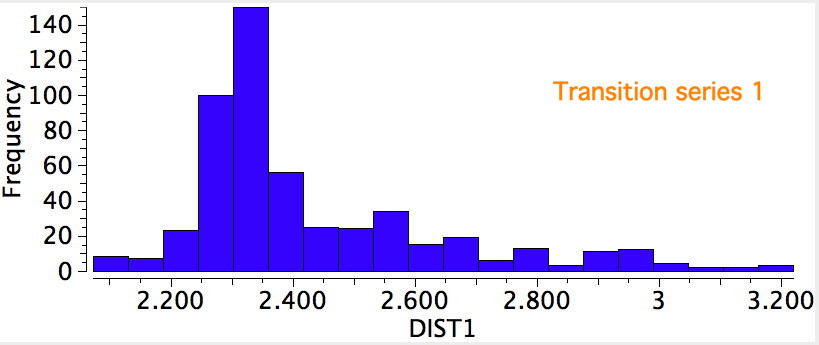
I noted previously that some 8-ring cyclic compounds could exist in either a planar-aromatic or a non-planar-non-aromatic mode, the mode being determined by apparently quite small changes in a ring substituent. Hunting for other examples of such chemistry on the edge, I did a search of the Cambridge crystal database for metal sulfides.
The butterfly effect summarises how a small change to a system may result in very large and often unpredictable (chaotic) consequences. If the system is merely on the edge of chaos, the consequences are predictable, but nevertheless finely poised between e.g. two possible outcomes. Here I ask how a molecule might manifest such behaviour.
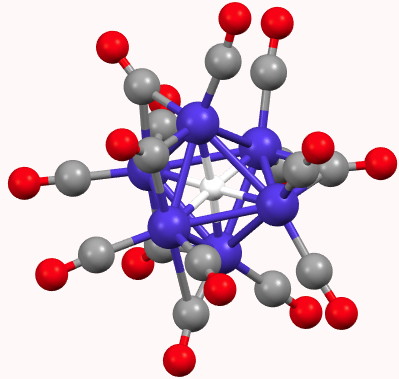
A feature of a blog which is quite different from a journal article is how rapidly a topic might evolve. Thus I started a few days ago with the theme of dicarbon (C2), identifying a metal carbide that showed C2 as a ligand, but which also entrapped a single carbon in hexa-coordinated mode.
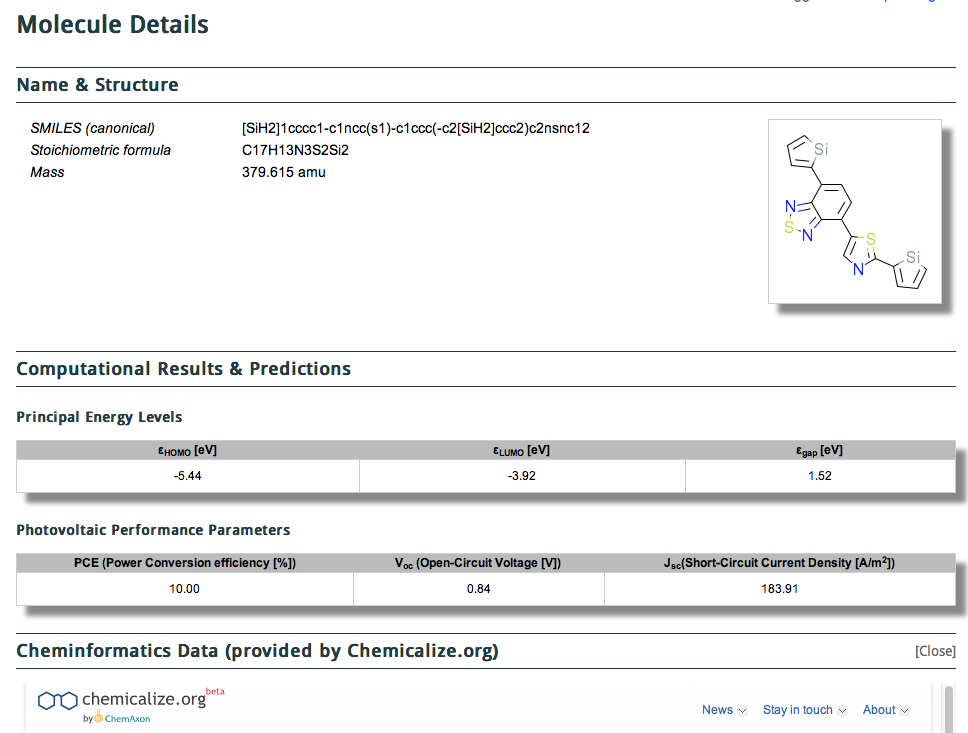
The title of this post summarises the contents of a new molecular database: www.molecularspace.org and I picked up on it by following the post by Jan Jensen at www.compchemhighlights.org (a wonderful overlay journal that tracks recent interesting articles). The molecularspace project more formally is called “The Harvard Clean Energy Project: Large-scale computational screening and design of organic […]
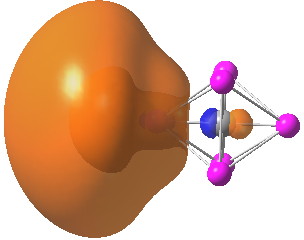
A comment made on the previous post on the topic of hexa-coordinate carbon cited an article entitled “Observation of hypervalent CLi6 by Knudsen-effusion mass spectrometry” by Kudo as a amongst the earliest of evidence that such species can exist (in the gas phase). It was a spectacular vindication of the earlier theoretical prediction, that such 6-coordinate […]
C2 (dicarbon) is certainly interesting from a theoretical point of view. Whether or not it can be described as having a quadruple bond has induced much passionate discussion,,,. Its occurrence in space and in flames is also well-known. But does it have what might be called a conventional chemistry?
A reader asked me about the mechanism of the reaction of 2-picoline N-oxide with acetic anhydride to give 2-acetoxymethylpyridine (the Boekelheide Rearrangement). He wrote ” I don’t understand why the system should prefer to go via fragmentation-recombination (… the evidence being that oxygen labelling shows scrambling) when there is an easy concerted pathway available (… a [3,3]sigmatropic shift). Furthermore, is […]
The blog post by Rich Apodaca entitled “The Horrifying Future of Scientific Communication” is very thought provoking and well worth reading. He takes us through disruptive innovation, and how it might impact upon how scientists communicate their knowledge.
A little while ago, I set out some interpretations of how to push curly arrows.

Many moons ago, when I was a young(ish) lecturer, and much closer in time to my laboratory roots of organic synthesis, I made some chemistry videos. One of these has resurfaced, somewhat (to me at least) unexpectedly.